 
Spectra is a fancy term for graphs that display light intensity versus wavelength. Atomic emission spectroscopy measures the intensity of light emitted by the excited atoms, while atomic absorption spectroscopy measures the light absorbed by atomic absorption. The light emitted by relaxing electrons produces atomic spectra. Which can be related to Einstein’s equationīoth atomic emission and atomic absorption spectroscopy can be used to analyze samples. Various orders of the spectrum are shown. Plank proposed radiation emitted energy in discrete packets (quanta), Figure 1: Schematic illustration of diffraction of light by a diffraction grating. For example, an atom in its lowest possible energy state (called the. This difficulty may be overcome by taking. A detector measures wavelengths of light which are absorbed or emitted by the hydrogen atoms. It relates the distribution to the thermal temperature of the system (as opposed to electronic temperature, vibrational temperature, or rotational temperature). Atomic energy levels are typically measured by observing transitions between two levels. For example, the intensity of the spectrum of a metal or alloy may vary with the metallurgical history of the sample. Light with many wavelengths is shone on the hydrogen sample. The Maxwell-Boltzmann equation gives the number of electrons in any given orbital. The resonance line is then defined as the specific radiation absorbed to reach the excited state. The energies of the various stationary states, or restricted orbits, can then be determined by these emission lines. When an atom is excited, the valence electron moves up an energy level. 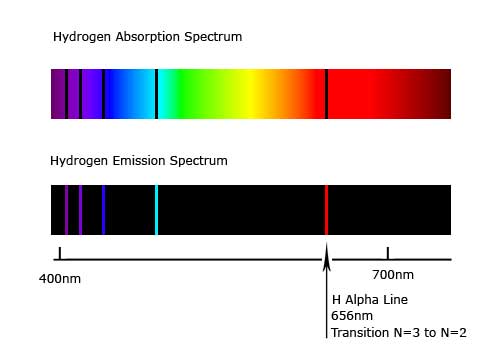 
Planck’s Contribution Energy is quantized in some systems, meaning that the system can have only certain energies and not a continuum of energies, unlike the classical case. Atoms can be excited when irradiated, which creates an absorption spectrum. Explain why atomic spectra indicate quantization. The atomic absorption lines show a deviation from a Lorentzian line-shape due to energy dependent matrix elements of the localized electronic state coupling to the photon field. For example, if an electron in a hydrogen atom makes a transition from the n 3 to the n 2 shell, the atom emits a photon with a wavelength c f h c h f h c E h c E 3 E 2, 8.36 where E E 3 E 2 is energy carried away by the photon and h c 1240 eV nm. \): Austrian physicist Wolfgang Pauli (1900 - 1958).Ītoms have valence electrons, which are the outermost electrons of the atom. We present a method to calculate the natural line width and energy dependent line shape due to fluorescence decay of core excited atoms within a full relativistic multi-reference configuration interaction theory. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
